Stents Market Size and Share
Stents Market Analysis by ���ϲ�����
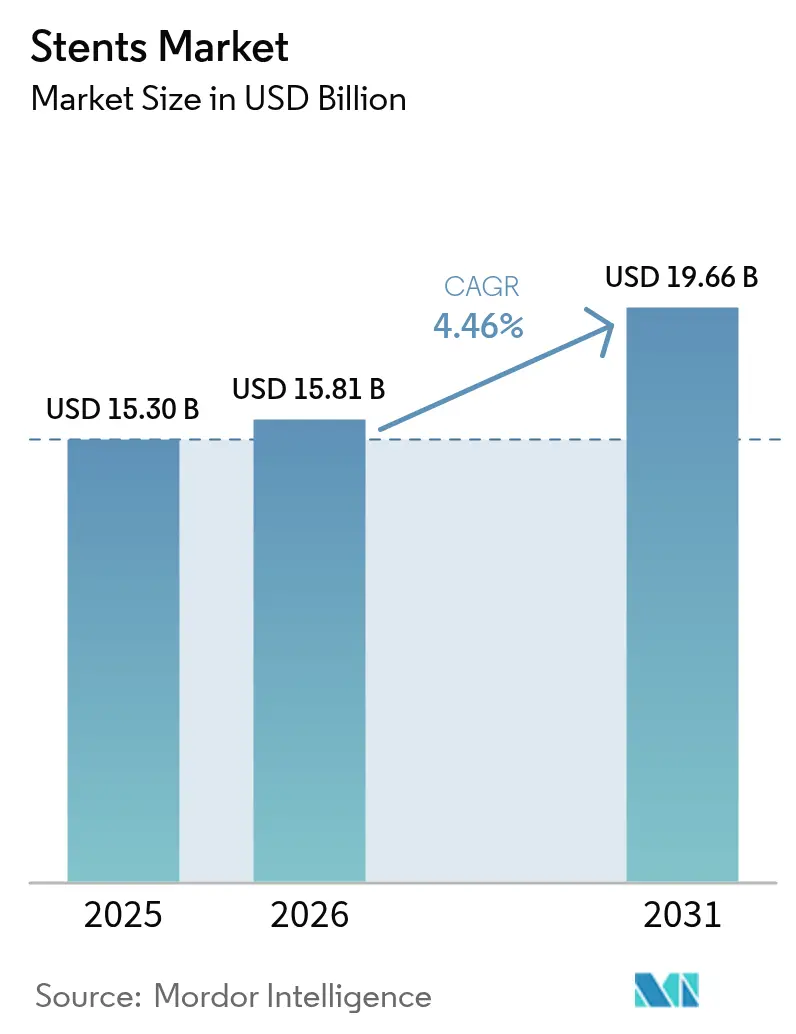
The Stents Market size is expected to grow from USD 15.30 billion in 2025 to USD 15.81 billion in 2026 and is forecast to reach USD 19.66 billion by 2031 at 4.46% CAGR over 2026-2031.
Growing procedure volumes in Asia-Pacific, rapid uptake of AI-guided robotic navigation, and renewed confidence in bioresorbable scaffolds are sustaining steady expansion even as global price ceilings pressure margins. Coronary devices dominate today, yet neurovascular flow-diverter and peripheral self-expandable platforms are emerging as the next wave of growth. Materials innovation is accelerating, with thinner cobalt-chromium struts and polymeric resorbable backbones reducing late adverse events. Simultaneously, reimbursement reforms that reward outpatient percutaneous coronary intervention (PCI) are moving a share of implantations from hospitals to ambulatory surgical centers, realigning revenue across the value chain.
Key Report Takeaways
- Coronary stents accounted for 61.46% of the 2025 stents market share, while neurovascular devices are forecast to post the fastest 8.46% CAGR through 2031.
- Metallic biomaterials held 63.68% of the 2025 mix; polymeric scaffolds are projected to rise at a 7.56% CAGR as bioresorbable platforms gain traction.
- Balloon-expandable delivery captured 57.82% revenue in 2025, but self-expandable formats are expected to advance at an 8.31% CAGR as peripheral disease interventions climb.
- Hospitals retained 69.22% end-user revenue in 2025, yet ambulatory surgical centers lead growth with a projected 7.24% CAGR to 2031 on the back of same-day-discharge PCI.
- North America generated 32.58% of 2025 sales; Asia-Pacific is anticipated to expand at a 6.36% CAGR, driven by high-volume, low-price procurement in China and India.
Note: Market size and forecast figures in this report are generated using ���ϲ�����’s proprietary estimation framework, updated with the latest available data and insights as of 2026.
Global Stents Market Trends and Insights
Drivers Impact Analysis*
| Driver | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Rising Prevalence of Cardiovascular & Peripheral Arterial Diseases | +0.9% | Global, highest in North America, Europe, urban Asia-Pacific | Long term (≥4 years) |
| Growing Demand for Minimally Invasive Percutaneous Interventions | +0.7% | Global, led by North America and Western Europe | Medium term (2-4 years) |
| Technological Advancements in Drug-Eluting & Bioresorbable Stents | +0.6% | North America, Europe, Japan, South Korea | Medium term (2-4 years) |
| Outpatient PCI Expansion Via ASC-Friendly Reimbursement Reforms | +0.5% | United States, select European markets | Short term (≤2 years) |
| AI-Enabled Imaging & Robotic Navigation Accelerating Adoption | +0.4% | North America, Western Europe, Japan | Medium term (2-4 years) |
| Rapid Roll-Out of Budget-Priced, Government-Capped Stents in India & China | +0.8% | India, China, Southeast Asia | Short term (≤2 years) |
| Source: ���ϲ����� | |||
Rising Prevalence of Cardiovascular & Peripheral Arterial Diseases
Cardiovascular disorders caused 17.9 million deaths in 2024, equal to 32% of all global mortality, elevating demand for revascularization tools such as stents.[1]World Health Organization, “Cardiovascular Diseases (CVDs),” WHO, who.int Diabetes affected 537 million adults in 2025, accelerating atherosclerotic plaque progression and necessitating repeat interventions. In the United States, coronary artery disease affected 20.5 million adults in 2024, with annual PCI procedures surpassing 1 million.[2]American Heart Association, “Heart Disease and Stroke Statistics — 2024 Update,” AHA, heart.orgPeripheral arterial disease touches 200 million people worldwide, but treatment rates remain below 15% in low-income nations, suggesting untapped pent-up need. Aging populations in Japan and Western Europe are presenting more complex multivessel disease, prompting thinner-strut device design. Emerging data also link prolonged exposure to PM2.5 above 100 µg/m³ in parts of India and China to a 30% surge in acute coronary syndrome admissions.
Growing Demand for Minimally Invasive Percutaneous Interventions
Patients favor catheter-based therapy over surgical bypass owing to shorter stays and quicker recovery. Same-day discharge reached 22% of U.S. elective PCI cases in 2025, up from 8% in 2020, aided by radial access and refined anticoagulation.[3]Society for Cardiovascular Angiography and Interventions, “Same-Day Discharge After Elective PCI,” SCAI, scai.org Drug-eluting stents now secure 18-month patency above 80% in diabetic superficial femoral artery cohorts, broadening peripheral indications. Neurovascular thrombectomy with adjunct flow-diverter implantation is reducing stroke disability when performed within a six-hour window. Outpatient migration is trimming per-case reimbursement, compelling manufacturers to negotiate volume-based contracts. The European Medicines Agency has fast-tracked devices incorporating novel polymers to cut required dual-antiplatelet therapy durations.
Technological Advancements in Drug-Eluting & Bioresorbable Stents
Next-generation coronary devices employ biodegradable coatings that release sirolimus-class agents over 3-6 months and then dissolve, easing long-term inflammation. Abbott re-launched Absorb trials in 2024 with optimized poly-L-lactic acid that fully hydrolyzes in 24 months. Boston Scientific’s polymer-free Eluvia won FDA clearance in 2025, recording late-lumen-loss of just 0.09 mm at one year. Ultra-thin (<70 µm) cobalt-chromium struts improve deliverability without sacrificing radial force. Optical coherence tomography reveals angiography-guided implants are malapposed in 18% of cases, so vendors now bundle imaging consoles with stent sales.
Outpatient PCI Expansion Via ASC-Friendly Reimbursement Reforms
CMS added elective PCI to the ASC list in January 2024 with a national payment of USD 8,200, catalyzing a wave of 47 new cardiovascular centers in 2025. Facilities apply CathPCI risk scores to triage candidates, excluding those with left-ventricular ejection fraction below 30%. Radial access plus bivalirudin drives vascular complication rates below 1%, enabling walk-out discharge. Major private insurers rapidly mirrored CMS policy. Profit-sharing has encouraged physician-owned centers, redistributing facility fees away from hospitals.
Restraints Impact Analysis*
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Stringent Regulatory Approval & Post-Market Surveillance Demands | -0.5% | Global, most stringent in United States, EU, Japan | Long term (≥4 years) |
| In-Stent Thrombosis / Restenosis Risks & Related Litigation | -0.3% | North America, Europe | Medium term (2-4 years) |
| Supply-Chain Fragility for Cobalt-Chromium & Nitinol Alloys | -0.4% | Global, acute in Asia-Pacific manufacturing hubs | Short term (≤2 years) |
| Rising Uptake of Drug-Coated Balloons & Intravascular Lithotripsy | -0.6% | Europe, United States, Japan | Medium term (2-4 years) |
| Source: ���ϲ����� | |||
Stringent Regulatory Approval & Post-Market Surveillance Demands
The FDA now requires five-year data on 1,500 patients for drug-eluting stent approvals, doubling timelines to 36 months and elevating per-device trial costs to USD 40 million. EU MDR enforcement since May 2024 mandates annual factory audits, producing a 14-month CE-renewal backlog. Japan’s PMDA permits conditional entry for bioresorbable scaffolds after two-year data but insists on manufacturer-funded registries. Administrative load favors incumbents with deep regulatory teams and deters venture-backed entrants.
In-Stent Thrombosis / Restenosis Risks & Related Litigation
Meta-analyses published in 2024 cited a 0.6% annual late-thrombosis rate for second-generation drug-eluting stents, equating to 6,000 U.S. events yearly. Dual-antiplatelet adherence drops to 62% at 18 months, amplifying risk. Class-action suits filed in 2025 over first-generation paclitaxel devices seek USD 200 million in damages. Neointimal hyperplasia drives 8-12% restenosis rates within two years, prompting costly repeat interventions. Regulators now direct manufacturers to embed thrombosis risk calculators in labeling.
*Our updated forecasts treat driver/restraint impacts as directional, not additive. The revised impact forecasts reflect baseline growth, mix effects, and variable interactions.
Segment Analysis
By Stent Type: Neurovascular Expansion Outpaces Coronary Maturity
Coronary devices retained 61.46% of the 2025 stents market share on the strength of entrenched use in ischemic heart disease. Growth, however, is leveling as drug-eluting penetration tops 90% and bare-metal applications shrink to graft lesions. Neurovascular stents are forecast to post an 8.46% CAGR through 2031, propelled by the widespread rollout of flow-diverter technology that safely treats wide-neck aneurysms without open surgery. Peripheral vascular implants benefit from an endovascular-first approach to limb salvage, with patency above 75% at three years when combined with drug-coated balloons.
Neurovascular momentum underscores a broader shift in the stents market toward complex, high-acuity indications, where reimbursement per case remains robust despite pricing pressure elsewhere. Gastrointestinal, urological, and airway segments together account for a small slice of the overall stents market size but are advancing rapidly as biodegradable polymers eliminate retrieval needs in palliative oncology settings. Bioresorbable coronary scaffolds, although still trial-restricted, are expected to re-enter commercialization on refined polymer chemistry backed by the ABSORB IV data set due in 2026.


By Material: Polymeric Platforms Tackle Long-Term Complications
Metallic alloys such as cobalt-chromium and platinum-chromium commanded 63.68% of 2025 revenue because they offer thin struts without sacrificing radial strength. Polymeric constructs are projected to climb at a 7.56% CAGR on rising surgeon confidence that resorbable devices reduce late thrombosis. Within that niche, poly-L-lactic acid dominates but newer blends featuring polycaprolactone are tuning degradation to 18-24 months, restoring vasomotion once support is no longer needed.
Interest in natural biomaterials such as collagen coatings remains modest, yet they serve patients with metal hypersensitivity and pediatric congenital defects requiring temporary scaffolds. Polymer-free drug-eluting stents provide a tactical intermediate, keeping metallic reliability while dropping the inflammatory load of carrier polymers. This transition illustrates how the stents market size growth intertwines with material science breakthroughs.
By Mode of Delivery: Self-Expandable Designs Climb in Tortuous Anatomy
Balloon-expandable platforms led with 57.82% revenue in 2025 thanks to precise inflation control in rigid, calcified coronary lesions. Self-expandable nitinol models are forecast to outpace at an 8.31% CAGR as peripheral and neurovascular work shifts volume their way. These devices can recover a pre-programmed diameter after compression and ride out vessel flexion, a critical trait in femoral arteries experiencing 40% cyclical diameter change during walking.
Carotid and renal segments are transitioning toward self-expandable architectures that maintain wall apposition as vessels remodel. Flow-diverter constructs, essentially tightly braided self-expandable mesh, redirect blood away from aneurysm sacs while keeping side branches patent. Hybrid balloon-plus-self designs for aortic bifurcations are being prototyped, though regulatory guidance is still emerging.

By End User: Ambulatory Centers Capture Same-Day Discharge Momentum
Hospitals continued to hold 69.22% of procedure revenue in 2025 because they treat multivessel and high-risk patients requiring surgical backup. Ambulatory surgical centers are predicted to rise at a 7.24% CAGR, energized by CMS policy and patient preference for shorter stays. Radial access provides sub-1% vascular complication rates, allowing six-hour observation windows.
Cardiac clinics—office-based labs with limited anesthesia—are eyeing single-vessel, stable lesions but remain constrained by state regulations that reserve complex interventions for hospitals. In dense metropolitan areas, the margin differential motivates clinicians to form physician-owned ASCs, recapturing facility fees previously ceded to hospitals. Overseas, Germany and the United Kingdom have begun pilot ASC PCI programs, though hospital-centric rules slow uptake.
Geography Analysis
North America led with 32.58% of 2025 revenue as sophisticated imaging suites, AI-enabling software, and premium bioresorbable launches kept average selling prices elevated. U.S. procedure counts are steady, but downward price renegotiations with group purchasing organizations are tempering top-line growth. Canada’s single-payer system is slower to adopt bioresorbables, yet provincial health authorities are funding robotics pilots to offset rural cardiologist shortages.
Asia-Pacific is projected to deliver a 6.36% CAGR to 2031 and will likely overtake Europe in absolute revenue by late forecast. China’s procurement program dropped unit prices to USD 97, propelling a 140% spike in volumes as rural cath-lab build-outs captured previously untreated cases. India performed 1.2 million coronary interventions in 2025 after sustained price caps lowered out-of-pocket costs below catastrophic thresholds. Southeast Asian ministries are drafting copycat tenders, signaling sustained double-digit unit growth even if revenue per case compresses.
Europe faces budget ceilings and growing preference for drug-coated balloons that cost 40% less than metallic drug-eluting stents. Germany, France, and the United Kingdom together make up 60% of regional consumption, yet price erosion dominates as national health services pursue bundled cardiology payments. Japan’s ultra-aged demographic is fueling peripheral and neurovascular demand, but biannual fee schedule reductions limit monetization. The Middle East and Africa leverage oil-funded investment in catheterization labs, but a dearth of trained interventionists keeps procedure density low. South America’s public-sector expansion lifts basic coronary volumes, although currency swings and tariff burdens complicate supply chains in Brazil and Argentina.

Competitive Landscape
The top five players include Abbott, Medtronic, Boston Scientific, Terumo, and B. Braun, signifying a moderately concentrated field. Multinationals defend margin by bundling robotic navigation, AI imaging, and ultra-thin strut technology into premium offerings, while regional challengers pursue a value-tier strategy anchored on government tenders at prices 40–50% below legacy levels. Vertical integration is expanding as Boston Scientific secured a 30% stake in a Chinese nitinol supplier in 2024 to hedge material volatility.
Patent filings remain vigorous; 1,247 stent-related applications reached the USPTO in 2025, focusing on polymer degradation kinetics, anti-thrombotic coatings, and OCT integration. Smaller entrants such as Elixir Medical and Translumina chase niche lesions like chronic total occlusions. The ascent of drug-coated balloons and lithotripsy compels portfolio diversification, with firms submitting combo-device approvals pairing calcium modification with resorbable scaffolds. Industry consensus anticipates a bifurcated market: premium innovation for complex disease in developed settings and cost-optimized, ISO-compliant workhorses for emerging economies.
Stents Industry Leaders
Boston Scientific Corporation
Medtronic PLC
Abbott Laboratories
Terumo Corporation
BIOTRONIK SE & Co. KG
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- February 2026: Parrish Medical Center performed Florida’s first U.S. implant of the Esprit BTK dissolvable stent for below-the-knee disease following FDA clearance
- January 2026: W. L. Gore & Associates secured FDA approval of the Viabahn Fortegra venous stent for IVC and iliofemoral disease.
- January 2026: Boston Scientific obtained Japanese MHLW approval for the PROMUS Everolimus-Eluting Coronary Stent System, pending reimbursement listing.
Research Methodology Framework and Report Scope
Market Definitions and Key Coverage
Our study treats the global stents market as the combined annual sales value of implantable mesh-like tubes designed to keep natural blood vessels or hollow organs open, embracing vascular devices (coronary, peripheral, neurovascular) alongside non-vascular formats such as gastrointestinal, airway, and urological stents. We consider only new, finished devices sold to healthcare providers or distributors and value them in USD at manufacturer selling price.
Scope exclusions include custom stent graft systems fabricated case-by-case for aortic aneurysm repair, dental and orthopedic placeholder implants, and refurbished or re-processed stents, which are excluded from the sizing.
Segmentation Overview
- By Stent Type
- Coronary Stents
- Drug-Eluting Coronary Stents
- Bare-Metal Coronary Stents
- Bioresorbable Coronary Scaffolds
- Peripheral Vascular Stents
- Iliac Artery Stents
- Femoral-Popliteal Stents
- Carotid Artery Stents
- Renal Artery Stents
- Neurovascular Stents
- Flow-Diverter Stents
- Intracranial Atherosclerotic Stents
- Gastrointestinal Stents
- Biliary Stents
- Esophageal Stents
- Colonic / Enteral Stents
- Urological Stents
- Ureteral Stents
- Prostatic Stents
- Airway Stents
- Tracheal Stents
- Bronchial Stents
- Coronary Stents
- By Material
- Metallic Biomaterials
- Polymeric Biomaterials
- Natural Biomaterials
- By Mode of Delivery
- Balloon-Expandable Stents
- Self-Expandable Stents
- By End User
- Hospitals
- Ambulatory Surgical Centers
- Cardiac & Specialty Clinics
- By Geography
- North America
- United States
- Canada
- Mexico
- Europe
- Germany
- France
- United Kingdom
- Italy
- Spain
- Rest of Europe
- Asia-Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia-Pacific
- Middle East & Africa
- GCC
- South Africa
- Rest of Middle East & Africa
- South America
- Brazil
- Argentina
- Rest of South America
- North America
Detailed Research Methodology and Data Validation
Primary Research
Mordor analysts interviewed interventional cardiologists, peripheral vascular surgeons, gastroenterologists, supply-chain managers, and regional distributors across North America, Europe, Asia-Pacific, and Latin America. These discussions clarified the current procedure mix, typical device mark-ups, the pace of bioresorbable adoption, and likely price-volume shifts, letting us adjust desk findings and validate growth drivers.
Desk Research
We began with published procedure statistics and trade data from sources such as the World Health Organization, OECD Health Stats, and Eurostat to pin down national interventional volumes and device import trends. Regulatory filings available through the US FDA 510(k) database, the EU's EUDAMED notices, and Japan's PMDA approvals helped us map product availability timelines and average selling prices. Additional context came from peer-reviewed journals (JACC, Lancet), leading cardiology associations, and hospital procurement portals. Paid datasets like D&B Hoovers and Dow Jones Factiva supplied company revenue splits and recent tender information. The sources cited here are illustrative; many more were reviewed to cross-verify figures and assumptions.
Market-Sizing & Forecasting
A top-down model starts with country-level angioplasty, neurovascular, and endoscopy procedure counts, which are then multiplied by stent utilization rates and blended ASPs. Results are pressure-tested through selective bottom-up checks such as sampled supplier revenues and hospital purchase audits. Key variables include PCI per million population, peripheral artery disease prevalence, elective endoscopy growth, average dual-therapy stent premiums, reimbursement revisions, and currency movements. Forecasts rely on multivariate regression that links stent uptake to disease incidence trends, aging index, and health-expenditure growth, with scenario analysis applied where device price caps or novel scaffold launches may swing demand.
Data Validation & Update Cycle
Outputs pass three analyst reviews, anomaly screens against parallel device categories, and variance checks versus prior editions. Models refresh each year; interim updates are triggered when major regulatory approvals, safety alerts, or reimbursement changes occur. Before release, a fresh validation call with at least one regional Key Opinion Leader ensures the narrative reflects the latest clinical reality.
Why Mordor's Stents Baseline Deserves Confidence
Published figures often diverge because firms pick different device mixes, pricing tiers, and refresh cadences. Our disciplined scope choice and annual reconfirmation of core variables curb that drift.
Key gap drivers include whether non-vascular stents are counted, how ASP erosion is projected, and the year chosen as the base. Some publishers lean on single-region shipment audits or roll forward older ASPs, while Mordor blends procedure data from 40+ countries with live price checks, yielding a balanced, ready-for-boardroom baseline.
Benchmark comparison
| Market Size | Anonymized source | Primary gap driver |
|---|---|---|
| USD 15.05 B (2025) | ���ϲ����� | - |
| USD 15.38 B (2025) | Regional Consultancy A | Uses inflation-indexed list prices, limited APAC country coverage |
| USD 11.40 B (2024) | Global Consultancy B | Excludes non-vascular stents and applies earlier base year |
| USD 12.61 B (2025) | Industry Association C | Focuses only on cardiovascular stents segment |
In short, by tying market value to verified procedure counts, contemporaneous ASPs, and clearly stated inclusions, ���ϲ����� delivers a dependable starting point for strategic planning, free from hidden scope shifts or aging assumptions.


Key Questions Answered in the Report
What is the projected revenue of the stents market in 2031?
The stents market is forecast to reach USD 19.66 billion by 2031, expanding at a 4.46% CAGR from 2026.
Which stent segment is expected to grow fastest through 2031?
Neurovascular stents are projected to post the highest 8.46% CAGR as flow-diverter technology gains traction in stroke care.
How are government price caps shaping purchasing trends in Asia?
India and China have enforced strict price ceilings and volume-based tenders, slashing unit prices to as low as USD 97 and driving triple-digit growth in procedure volumes.
Why are ambulatory surgical centers important to future implant volumes?
CMS added elective PCI to the ASC list in 2024, enabling same-day discharge and sparking a 47-center construction boom that will lift ASC stent placements at a 7.24% CAGR.
What material innovations are influencing long-term outcomes?
Polymeric bioresorbable scaffolds made from optimized poly-L-lactic acid blends dissolve after vessel healing, reducing inflammation and late thrombosis risk compared to permanent metal.
How is AI improving stent deployment accuracy?
AI-driven imaging systems predict optimal stent sizing with 92% concordance to experts and guide robotic platforms that cut operator radiation by 95%.
Page last updated on:









