North America Hemodynamic Monitoring Market Analysis by ���ϲ�����
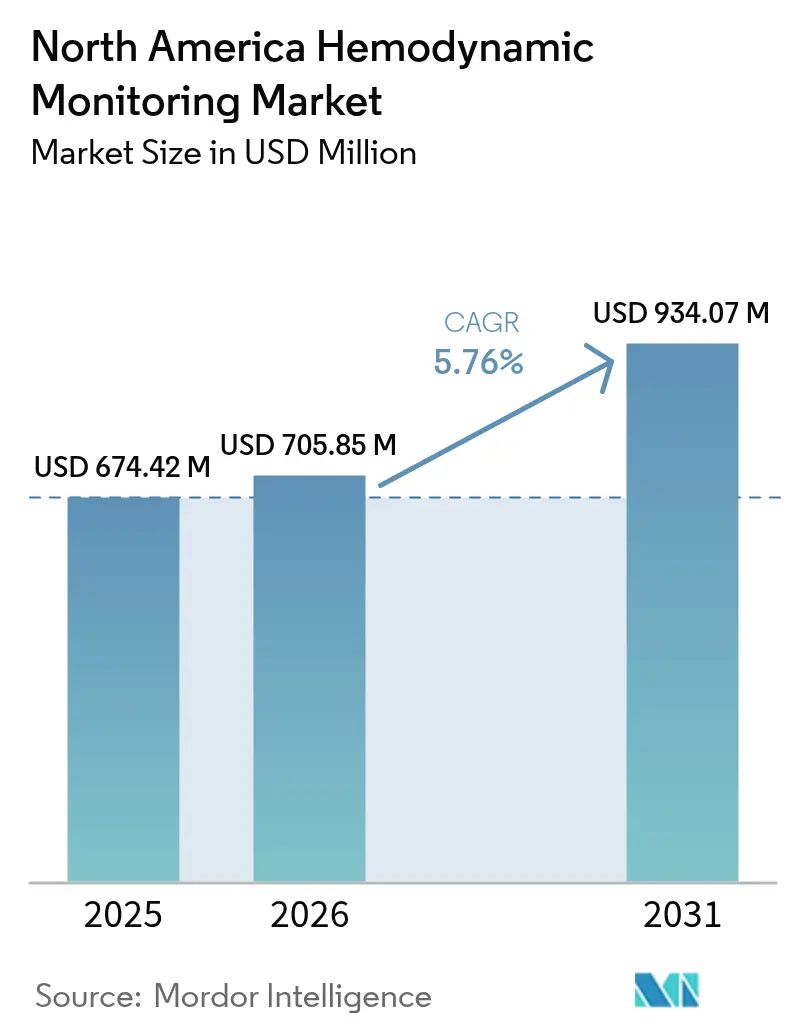
The North America Hemodynamic Monitoring Market size is expected to increase from USD 674.42 million in 2025 to USD 705.85 million in 2026 and reach USD 934.07 million by 2031, growing at a CAGR of 5.76% over 2026-2031.
Rising heart-failure prevalence, updated sepsis protocols, and reimbursement that now covers implantable wireless sensors are redirecting capital from catheter-based systems toward minimally invasive and non-invasive platforms. Hospitals increasingly invest in centralized dashboards that apply machine-learning to arterial-waveform data, while tele-ICU networks extend continuous monitoring to community facilities. Competition is intensifying as startups add wearable patches and AI-driven alarm filtering, challenging incumbents whose portfolios revolve around invasive hardware. Mexico’s hospital-modernization program and Canada’s tele-ICU expansion add geographic momentum, yet high equipment costs and staff shortages temper adoption in smaller centers.
Key Report Takeaways
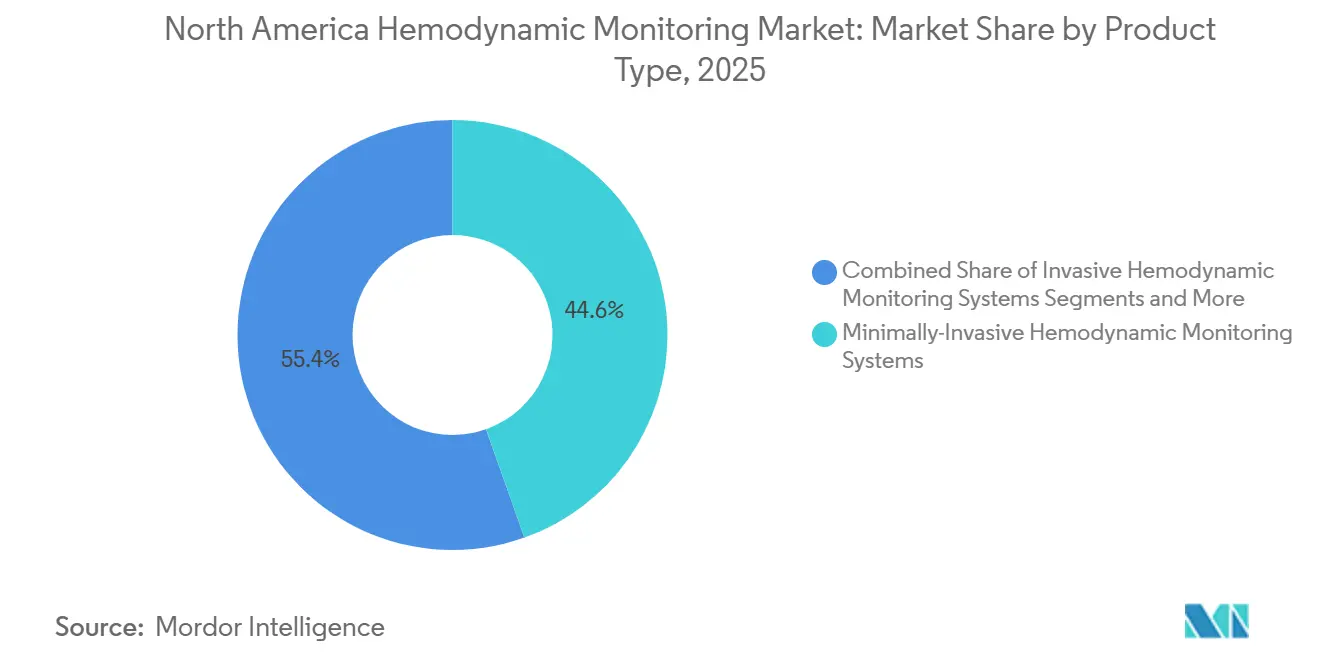
- Minimally invasive systems led with 44.56% of 2025 revenue, while non-invasive platforms are advancing at an 8.46% CAGR through 2031, the fastest in the product segment.
- Software and data-management platforms recorded the highest component-level growth, expanding at a 9.25% CAGR.
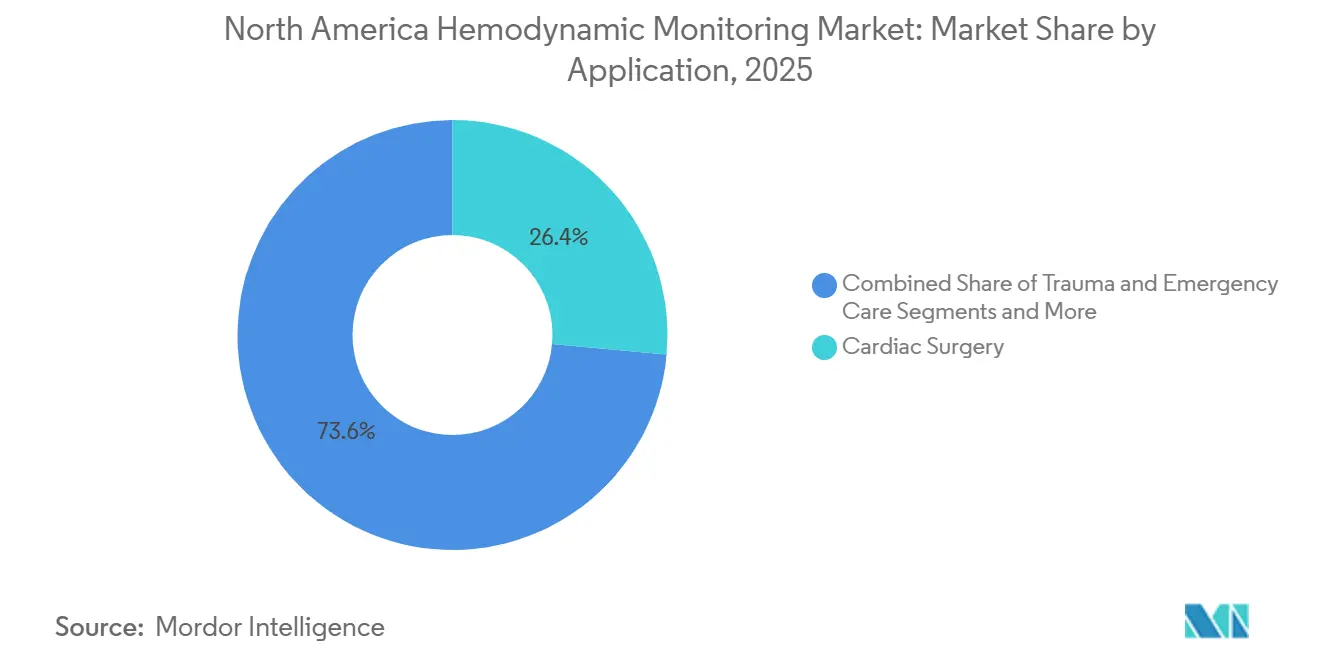
- Sepsis management is the quickest growing application, rising at a 7.73% CAGR, whereas cardiac surgery accounted for 26.44% of 2025 revenue.
- Tissue-oxygenation monitoring is projected to post an 8.23% CAGR, outpacing cardiac-output and blood-pressure parameters.
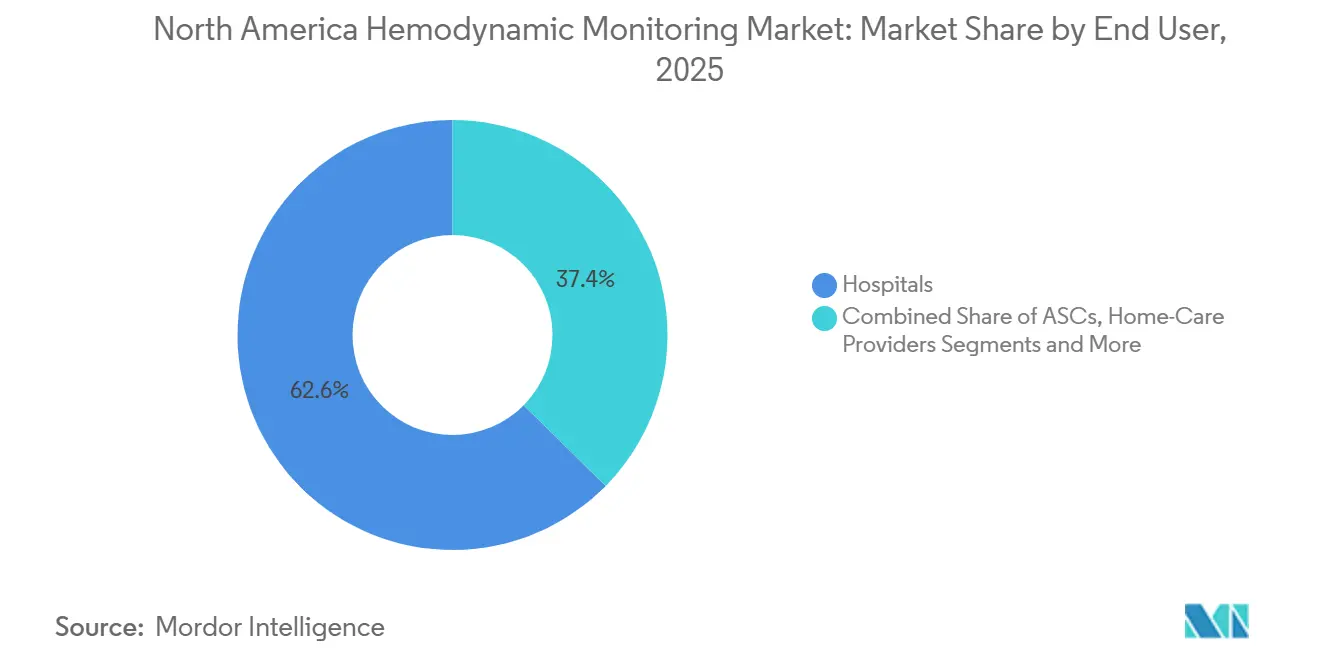
- Home-care providers held the fastest end-user trajectory at a 9.13% CAGR, although hospitals retained 62.62% of 2025 revenue.
- The United States contributed 76.78% of 2025 sales; Mexico is the regional growth leader at an 8.64% CAGR through 2031.
Note: Market size and forecast figures in this report are generated using ���ϲ�����’s proprietary estimation framework, updated with the latest available data and insights as of 2026.
North America Hemodynamic Monitoring Market Trends and Insights
Driver Impact Analysis*
| Driver | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| Rising Cardiovascular & Critical-Care Caseload | +1.2% | United States, Canada, spillover to Mexico | Long term (≥ 4 years) |
| Advances in Minimally Invasive Technologies | +1.4% | United States academic medical centers, regional diffusion | Medium term (2-4 years) |
| Rapidly Growing Elderly Population | +0.9% | United States, Canada | Long term (≥ 4 years) |
| Favorable U.S. Reimbursement Policies | +0.8% | United States | Short term (≤ 2 years) |
| AI-Guided Fluid-Management Analytics | +1.0% | United States, early use in Canada | Medium term (2-4 years) |
| Expansion of Tele-ICU Platforms | +0.7% | Rural United States and Canadian provincial networks | Medium term (2-4 years) |
| Source: ���ϲ����� | |||
Rising Cardiovascular & Critical-Care Caseload
Heart disease caused 702,880 U.S. deaths in 2022, and 6.7 million adults lived with heart failure in 2024; projections place the cohort at 8.7 million by 2030, extending ICU stays and favoring sensors that remain in-situ for days without infection risks.[1] Sepsis guidelines that mandate hemodynamic assessment within one hour further load ICUs, accelerating demand for pulse-contour and bioreactance monitors.[2] Mitchell M. Levy, “Surviving Sepsis Campaign: International Guidelines for Management of Sepsis and Septic Shock 2024,” Society of Critical Care Medicine, sccm.org
Advances in Minimally Invasive Monitoring Technologies
Fourth-generation FloTrac algorithms cleared in 2025 employ machine-learning corrections that narrow error margins during vasoplegic shock, while esophageal Doppler gained U.S. endorsement for guiding fluid therapy in major surgery.[3]U.S. Food and Drug Administration, “510(k) Premarket Notification K243781 – FloTrac System,” U.S. Food and Drug Administration, fda.gov Vendors that bundle sensors with cloud-connected decision support are displacing standalone hardware as clinicians prioritize workflow integration.
Rapidly Growing Elderly Population
Adults ≥ 65 years will reach 21.6% of the U.S. population by 2040, and the 85-plus group already consumes disproportionate ICU resources. Older cardiac-surgery patients exhibit longer hypotension episodes, driving continuous monitoring, and remote pulmonary-artery sensors now target chronic heart-failure management in this cohort.
Favorable U.S. Reimbursement Policies
CMS introduced coverage-with-evidence-development for implantable hemodynamic sensors in 2025, encouraging hospitals to collect outcomes data in return for payment. Commercial insurers mirrored the policy, reimbursing cardiologists USD 60-90 monthly for device-generated data review, which stimulates outpatient adoption.
Restraint Impact Analysis*
| Restraint | (~) % Impact on CAGR Forecast | Geographic Relevance | Impact Timeline |
|---|---|---|---|
| High Capital & Consumable Costs | -0.9% | U.S. community hospitals, Mexico public facilities | Short term (≤ 2 years) |
| Procedure-Related Infection & Complication Risks | -0.6% | North America, notably invasive catheter use | Medium term (2-4 years) |
| Clinician Alarm Fatigue & Data Overload | -0.5% | United States, Canada ICUs | Short term (≤ 2 years) |
| Shortage of Skilled Critical-Care Nurses | -0.4% | Rural United States and Canada | Long term (≥ 4 years) |
| Source: ���ϲ����� | |||
High Capital & Consumable Costs
A fully configured HemoSphere unit costs USD 35,000-50,000, and a 20-bed ICU may invest up to USD 600,000 before disposables; smaller hospitals struggle to justify this outlay, while Mexico’s IMSS budgets only USD 12,000 per ICU bed for all monitoring gear.
Procedure-Related Infection & Complication Risks
Central-line infections occur at 0.8 per 1,000 line-days and cost USD 45,000 per episode, while pulmonary-artery rupture carries 50% mortality, prompting clinicians to shift toward minimally invasive technologies.
*Our updated forecasts treat driver/restraint impacts as directional, not additive. The revised impact forecasts reflect baseline growth, mix effects, and variable interactions.
Segment Analysis
By Product Type: Non-Invasive Platforms Gain Share
Minimally invasive systems held 44.56% of 2025 revenue, anchored by pulse-contour and esophageal Doppler devices that avoid pulmonary-artery catheterization while supplying beat-to-beat data. However, non-invasive platforms are growing at an 8.46% CAGR through 2031 as tele-ICU models favor sensors that work without arterial lines. The North America hemodynamic monitoring market size for non-invasive platforms is expected to expand steadily under this momentum. Wearable patches under development correlate strongly with arterial-line measurements, suggesting future clearance will accelerate bedside replacement of invasive hardware.
The North America hemodynamic monitoring market share commanded by invasive catheters persists in complex cardiac cases, yet FDA approval of the Cordella pulmonary-artery sensor in 2024 validated ambulatory hemodynamics and pressured bedside-monitor vendors to match home-care capabilities. Vendors that integrate cloud connectivity, predictive analytics, and minimal consumable requirements are positioned to capture the next wave of procurement.

By Component: Software Platforms Accelerate
Monitors delivered 34.72% of 2025 component revenue, but software and data-management suites are advancing at a 9.25% CAGR, the quickest component trajectory. Hospitals regard predictive algorithms that alert clinicians 15 minutes before hypotension as essential rather than optional, redefining purchasing criteria. Open APIs that welcome third-party sepsis or ventilator-weaning algorithms differentiate platforms and encourage ecosystem stickiness.
Sensors and disposables still underpin recurring revenue, yet price pressure mounts as hospitals scrutinize total cost of ownership. The North America hemodynamic monitoring market size attributed to services remains stable because multi-year maintenance contracts accompany every capital sale. Vendors now emphasize analytic-software subscriptions to lock in annuity streams and offset hardware commoditization.
By Application: Sepsis Management Surges
Cardiac surgery accounted for 26.44% of 2025 revenue, thanks to high-acuity valve and bypass volumes that require real-time preload and afterload optimization. Updated Surviving Sepsis Campaign recommendations, however, propelled sepsis management to the fastest 7.73% CAGR, as dynamic fluid-responsiveness indices replace static pressure targets in emergency departments. Hospitals invest in bioreactance and pulse-contour devices to satisfy CMS’s SEP-1 quality measures, tying technology adoption directly to reimbursement retention.
General surgery and trauma applications continue to rely on non-invasive systems for goal-directed therapy, and interventional cardiology uses hemodynamics to monitor for tamponade and heart-failure flares. The North America hemodynamic monitoring market share linked to sepsis is projected to broaden as quality-metric compliance shapes purchasing decisions.

By Monitoring Parameter: Tissue Oxygenation Emerges
Cardiac output metrics led with 39.16% of 2025 parameter revenue, yet tissue oxygenation is projected to rise at an 8.23% CAGR. Near-infrared spectroscopy now tracks cerebral and somatic oxygen saturation during cardiac surgery, reducing postoperative stroke by 32% and delirium by 28% in 2024 trials. Regional perfusion insights complement systemic signals, giving clinicians an early warning of microcirculatory deficits that global pressures can miss.
Volume-status metrics, such as stroke-volume variation derived from pulse-contour analysis, anchor fluid-management protocols. Continuous blood-pressure monitoring remains ubiquitous, but algorithms that synthesize multi-parameter trends enhance interpretability and reduce false alarms, key aspirations for the North America hemodynamic monitoring market.
By End User: Home Care Accelerates
Hospitals retained 62.62% of 2025 revenue, yet home-care providers will see a 9.13% CAGR through 2031 as Medicare reimburses implantable sensor data reviews. Cordella-monitored patients recorded 38% fewer heart-failure admissions and saved the system USD 8,000-12,000 per patient annually, building a robust economic case.
Ambulatory surgical centers adopt minimally invasive devices selectively for high-risk cases, and specialty cardiology clinics leverage pulmonary-artery sensors for outpatient titration. Telehealth platforms that deliver actionable hemodynamic insights in the home setting promise to re-shape the North America hemodynamic monitoring market size allocation across care sites.

Geography Analysis
The United States generated 76.78% of 2025 revenue, buoyed by 95,000 ICU beds and a reimbursement framework that bundles monitoring into DRG payments. Tele-ICU beds grew to cover 18% of capacity in 2024, trimming ICU mortality by 11% and stay length by 0.8 days, justifying subscription fees and spurring additional sensor purchases. AI-driven platforms and wearable patches intensify vendor competition, but high product penetration has also increased price sensitivity among group purchasing organizations.
Canada contributes a smaller yet steady portion, anchored by provincial funding aimed at tele-ICU expansion. Ontario budgeted CAD 120 million (USD 88 million) for ICU upgrades in 2024, prioritizing pulse-contour and Doppler devices for fluid-guided therapy. Centralized purchasing slows rollouts, but once value is proven through CADTH assessments, adoption scales rapidly across provincial networks.
Mexico is advancing at an 8.64% CAGR, propelled by a USD 1.2 billion upgrade across IMSS hospitals that adds advanced monitoring to new ICUs. Diabetes prevalence at 16.9% swells cardiovascular load, yet high capital costs limit adoption outside private urban hospitals. Regulatory streamlining has cut device approval times to 9-12 months, but discount negotiations keep pricing pressure high, influencing vendor margin strategies within the North America hemodynamic monitoring market.
Competitive Landscape
Market concentration is moderate. Edwards Lifesciences, GE HealthCare, and Philips sustain strongholds through catheter and monitor franchises, but Masimo expands non-invasive territory with hemoglobin and perfusion analytics. Abbott’s Cordella system opened the home-monitoring frontier, while startups such as Gauss Surgical tackle alarm-noise reduction with AI. Software dominance is the new battleground: Edwards filed 14 machine-learning patents in 2024, and Masimo submitted nine covering fluid-responsiveness algorithms. Vendors offering open APIs win hospital favor by enabling integration of third-party sepsis predictors and ventilator-weaning tools, differentiators that pure-hardware manufacturers lack.
White-space opportunities cluster around home-based heart-failure care, AI-guided decision support, and tele-ICU compatible non-invasive systems. FDA guidance on SaMD tightened clearance requirements yet validated the clinical value of algorithmic platforms, giving data-rich incumbents a protective moat while raising entry costs for newcomers. Overall, suppliers must pivot from hardware specs to end-to-end clinical solutions to defend share in the evolving North America hemodynamic monitoring market.
North America Hemodynamic Monitoring Industry Leaders
Getinge AB
GE Healthcare
Koninklijke Philips N.V.
BD
ICU Medical
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- January 2026: eMurmur received FDA clearance for Heart AI, which identifies murmurs and derives hemodynamic insights from digital-stethoscope recordings.
- July 2025: Cardiosense secured 510(k) clearance for CardioTag, the first wearable that captures ECG, PPG, and SCG signals concurrently for broad cardiac-function assessment.
- April 2025: BD launched an advanced monitoring platform that uses AI algorithms to predict blood-pressure instability during procedures, aiming to avert life-threatening events early.
North America Hemodynamic Monitoring Market Report Scope
As per the scope of the report, Hemodynamic monitoring devices are tools used to measure heart function and circulatory system efficiency in real-time, aiding in assessing tissue perfusion, detecting instabilities, and guiding therapy for critically ill patients.
The North America Hemodynamic Monitoring Market Report is segmented by Product Type, Component, Application, Monitoring Parameter, End User, and Geography. By Product Type, the market is segmented into Invasive, Minimally Invasive, and Non-Invasive Systems. By Component, the market is segmented into Monitors, Sensors, Disposables, Software, and Services. By Application, the market is segmented into Cardiac Surgery, General Surgery, Interventional Cardiology, Trauma, and Sepsis. By Monitoring Parameter, the market is segmented into Cardiac Output, Blood Pressure, Volume Status, and Tissue Oxygenation. By End User, the market is segmented into Hospitals, ASCs, Home Care, and Specialty Clinics. By Geography, the market is segmented into the United States, Canada, and Mexico. Market Forecasts are Provided in Terms of Value (USD).
| Invasive Hemodynamic Monitoring Systems | Catheters |
| Pressure Transducers | |
| Consumables (pressure tubing, flush sets, etc.) | |
| Minimally-Invasive Hemodynamic Monitoring Systems | Pulse-Contour Analysis Devices |
| Esophageal Doppler Monitors | |
| Non-Invasive Hemodynamic Monitoring Systems | Impedance/ Bioreactance Cardiology Systems |
| Wearable Continuous-BP & CO Patch Sensors |
| Monitors |
| Sensors / Transducers |
| Disposables & Accessories |
| Software and Platforms |
| Services |
| Cardiac Surgery |
| General & Orthopedic Surgery |
| Interventional Cardiology |
| Trauma & Emergency Care |
| Sepsis and Septic Shock Management |
| Cardiac Output & Derived Indices (CI, SV, SVV, PPV) |
| Continuous Blood Pressure (ART/NIBP) |
| Volume Status / Fluid Responsiveness Metrics |
| Tissue Oxygenation (StO₂, rSO₂) |
| Hospitals |
| Ambulatory Surgical Centers |
| Home-Care Providers |
| Specialty Cardiology & Critical-Care Clinics |
| United States |
| Canada |
| Mexico |
| By Product Type | Invasive Hemodynamic Monitoring Systems | Catheters |
| Pressure Transducers | ||
| Consumables (pressure tubing, flush sets, etc.) | ||
| Minimally-Invasive Hemodynamic Monitoring Systems | Pulse-Contour Analysis Devices | |
| Esophageal Doppler Monitors | ||
| Non-Invasive Hemodynamic Monitoring Systems | Impedance/ Bioreactance Cardiology Systems | |
| Wearable Continuous-BP & CO Patch Sensors | ||
| By Component | Monitors | |
| Sensors / Transducers | ||
| Disposables & Accessories | ||
| Software and Platforms | ||
| Services | ||
| By Application | Cardiac Surgery | |
| General & Orthopedic Surgery | ||
| Interventional Cardiology | ||
| Trauma & Emergency Care | ||
| Sepsis and Septic Shock Management | ||
| By Monitoring Parameter | Cardiac Output & Derived Indices (CI, SV, SVV, PPV) | |
| Continuous Blood Pressure (ART/NIBP) | ||
| Volume Status / Fluid Responsiveness Metrics | ||
| Tissue Oxygenation (StO₂, rSO₂) | ||
| By End User | Hospitals | |
| Ambulatory Surgical Centers | ||
| Home-Care Providers | ||
| Specialty Cardiology & Critical-Care Clinics | ||
| By Country | United States | |
| Canada | ||
| Mexico | ||


Key Questions Answered in the Report
How large is the North America hemodynamic monitoring market in 2026?
It stood at USD 705.85 million in 2026 and is projected to reach USD 934.07 million by 2031.
Which product type grows fastest through 2031?
Non-invasive platforms, expanding at an 8.46% CAGR as tele-ICU and home-care usage rises.
Why is sepsis management a key growth application?
Updated guidelines require continuous cardiac-output monitoring early in treatment, driving a 7.73% CAGR for sepsis-focused deployments.
How are AI tools changing hemodynamic monitoring?
Predictive algorithms now forecast hypotension up to 15 minutes in advance, reducing complications and guiding fluid therapy.
Which country offers the highest growth rate in the region?
Mexico, advancing at an 8.64% CAGR due to hospital modernization and a high diabetes-related cardiac burden.
What is the main restraint on market adoption?
High capital and consumable costs, especially for community hospitals and public facilities, limit widespread deployment.
Page last updated on:








