Defibrillator Market Size and Share
Defibrillator Market Analysis by ���ϲ�����
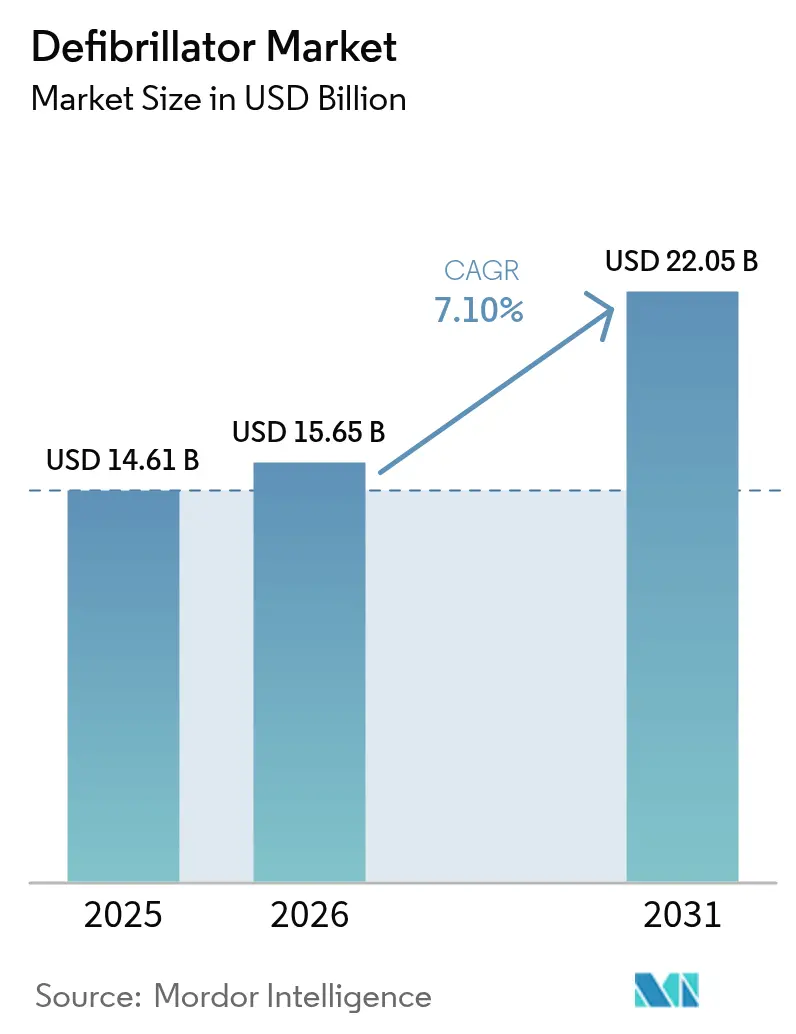
The Defibrillator Market size was valued at USD 14.61 billion in 2025 and is estimated to grow from USD 15.65 billion in 2026 to reach USD 22.05 billion by 2031, at a CAGR of 7.10% during the forecast period (2026-2031).
Sustained incidence of sudden cardiac arrest, rapid adoption of AI-enabled devices, and wider public-access programs keep demand resilient despite supply-chain headwinds. Device makers are lengthening battery life, embedding cloud connectivity, and leaning on predictive analytics to differentiate. Payers and regulators in high-income countries increasingly reimburse cloud monitoring, while emerging markets boost baseline healthcare outlays to narrow treatment gaps. Competitive intensity rises as innovators secure FDA clearances for extravascular implantation and patch-based wearables, signaling a new product cycle poised to accelerate the defibrillator market over the next five years.
Key Report Takeaways
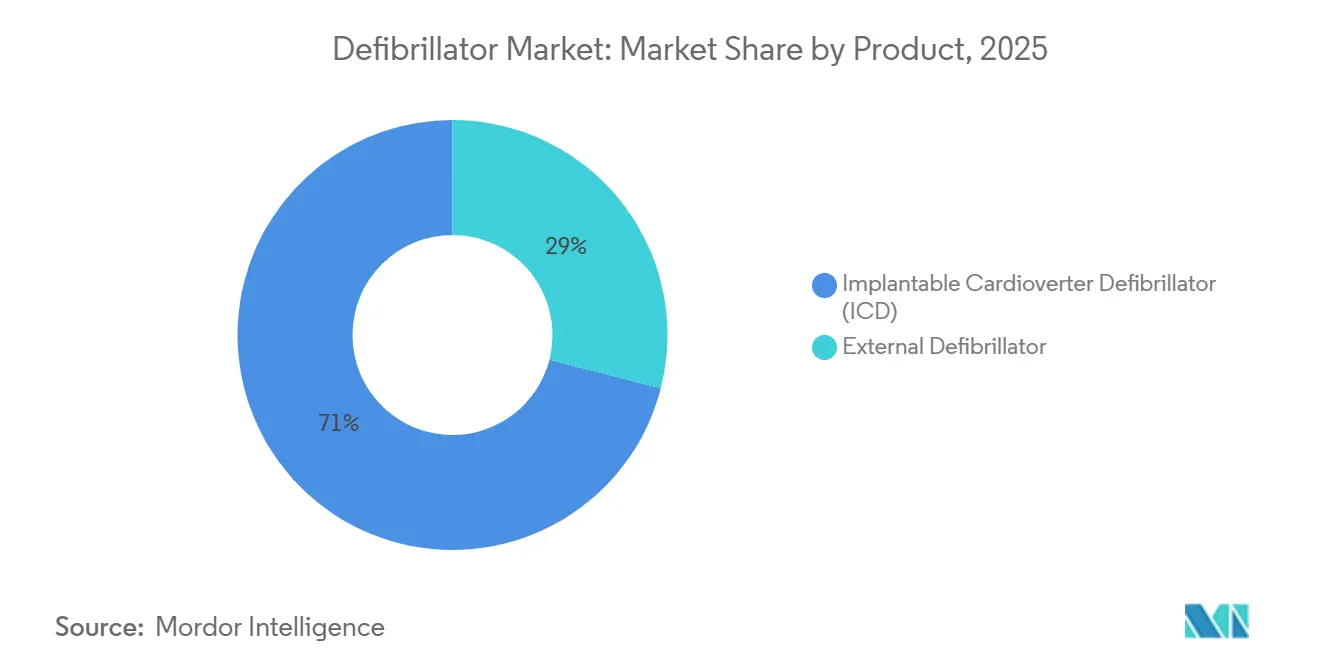
- By product category, implantable cardioverter defibrillators held 71.02% of defibrillator market share in 2025, while external defibrillators are projected to expand at a 7.64% CAGR to 2031.
- By end user, hospitals and cardiac centers accounted for 77.60% of the defibrillator market in 2025; home-care settings are projected to record the highest CAGR at 7.98% through 2031.
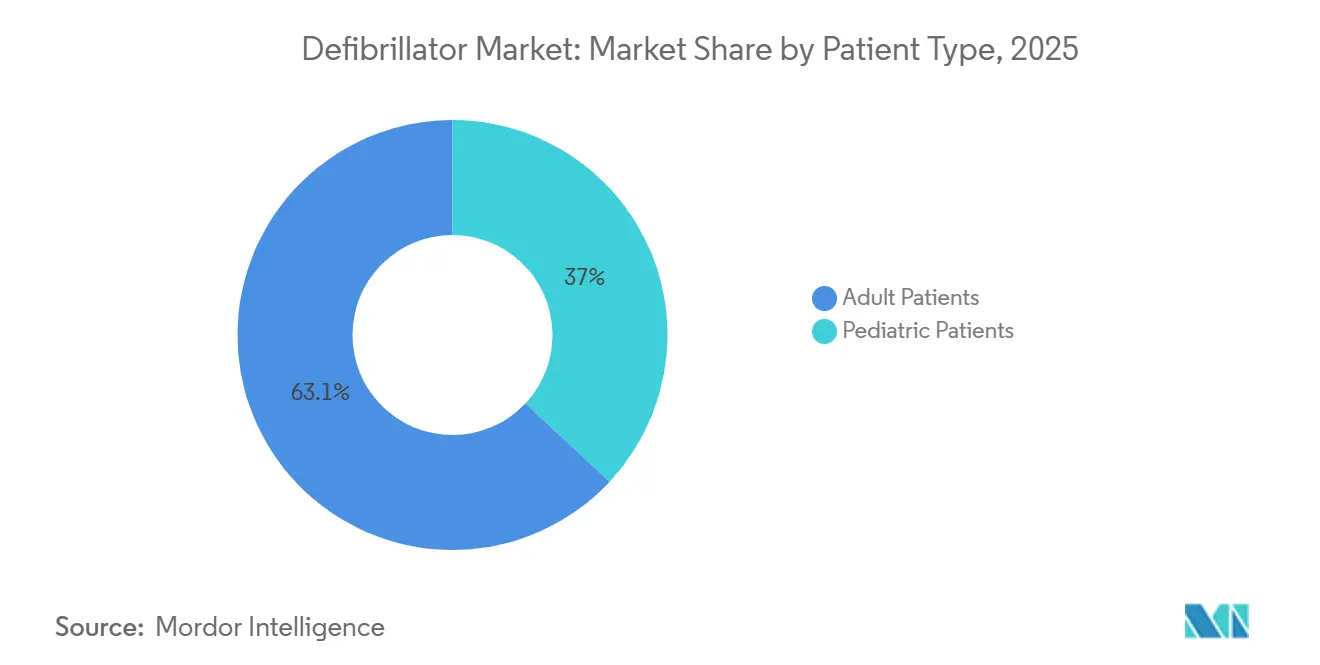
- By patient type, adult patients captured 63.05% share of the defibrillator market size in 2025, whereas pediatric applications are advancing at a 7.78% CAGR over the same horizon.
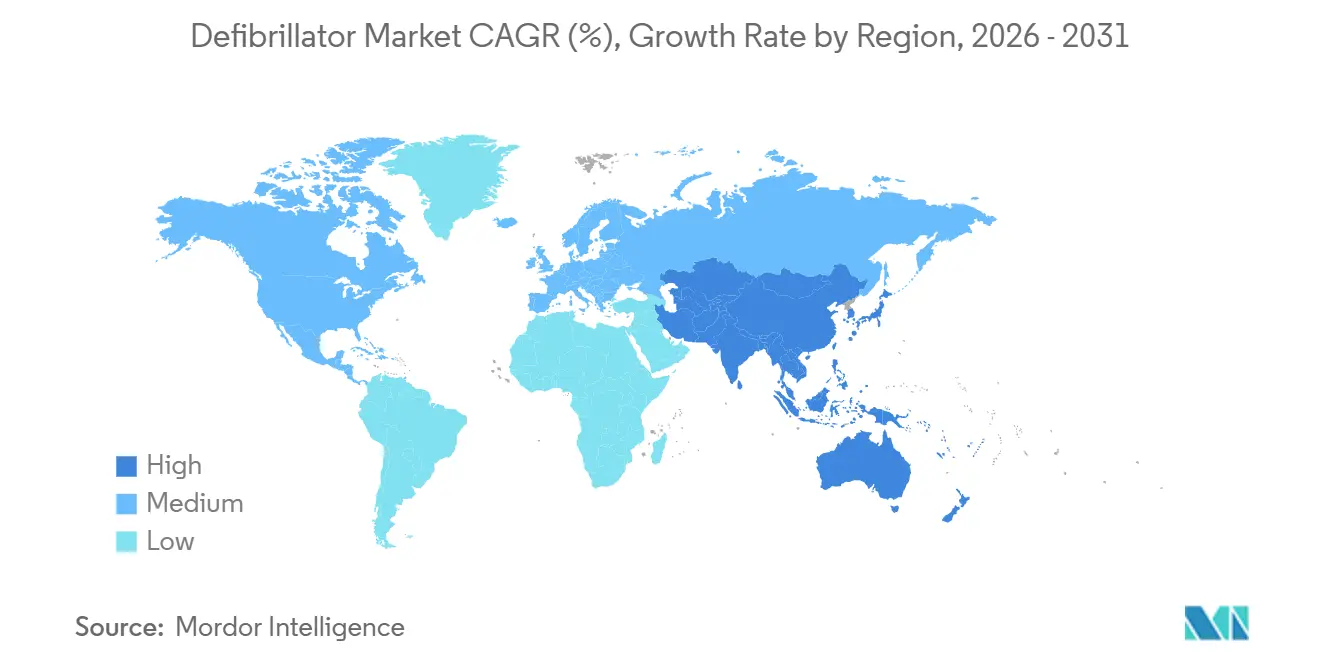
- By geography, North America commanded 43.85% of the defibrillator market share in 2025; Asia-Pacific is forecast to grow fastest at an 8.21% CAGR to 2031.
Note: Market size and forecast figures in this report are generated using ���ϲ�����’s proprietary estimation framework, updated with the latest available data and insights as of 2026.
Global Defibrillator Market Trends and Insights
Drivers Impact Analysis*
| DRIVER | (~) % IMPACT ON CAGR FORECAST | GEOGRAPHIC RELEVANCE | IMPACT TIMELINE |
|---|---|---|---|
| Rising prevalence of cardiovascular diseases | +1.8% | Global, highest absolute growth in APAC & MEA | Long term (≥ 4 years) |
| Technological advancements in ICDs and AEDs | +1.5% | North America & EU lead, APAC lags 18-24 months | Medium term (2-4 years) |
| Expansion of public-access defibrillation programs | +1.2% | Strongest in EU, variable in North America | Medium term (2-4 years) |
| Aging workforce training & simulation demand | +0.6% | North America & EU | Short term (≤ 2 years) |
| Subscription-based cloud-connected models | +0.7% | North America early, EU pilots, limited APAC | Medium term (2-4 years) |
| Drone-delivered AED networks | +0.4% | Nordics operational, U.S. pilots, APAC exploratory | Long term (≥ 4 years) |
| Source: ���ϲ����� | |||
Rising Prevalence of Cardiovascular Diseases
Cardiovascular disease (CVD) continues to be the top cause of mortality in the U.S., leading to over 940,000 deaths annually. By 2050, projections suggest that more than 60% of American adults may grapple with some form of CVD, a rise from the current 50%.[1]American Heart Association, “2026 Heart and Stroke Statistics,” heart.org In 2025, updated appropriate-use criteria expanded the candidacy for ICDs to include patients with non-ischemic cardiomyopathy and left-ventricular ejection fractions ranging from 35% to 40%. In alignment with these guidelines, Medicare introduced new reimbursement codes in 2025, potentially benefiting an additional 180,000 eligible beneficiaries. These combined clinical and reimbursement shifts are broadening the market for both implantable and external devices, bolstering the Defibrillator market. Meanwhile, India and China, grappling with rapid urbanization and dietary changes, are witnessing a swift rise in CVD prevalence, with hypertension rates surpassing 30% in major urban centers.
Technological Advancements in ICDs and AEDs
FDA approval of Medtronic’s Aurora EV-ICD in 2023 shifted the defibrillator market toward extravascular implantation, which avoids venous complications while delivering antitachycardia pacing, achieving 98.7% effectiveness.[2]Medtronic plc, “Medtronic Receives FDA Approval for Extravascular Defibrillator,” medtronic.com Battery longevity extends about 60%, improving lifetime economics for providers and patients. AI algorithms now curtail false alarms; Element Science’s Jewel Patch wearable won clearance in 2025 after demonstrating high compliance and low inappropriate-shock rates. Modular architectures are gaining traction, with Boston Scientific reporting 97.5% complication-free performance for its leadless pacemaker/defibrillator combination. Collectively, these innovations reinforce premium pricing and stimulate replacement demand, supporting mid-single-digit unit growth across the defibrillator market.
Expansion of Public-Access Defibrillation Programs
Jurisdictions mandate broader automated external defibrillator (AED) deployment, propelling the defibrillator market into non-clinical venues. All 50 U.S. states had supporting statutes by 2017, and new rules continue: Washington now obliges fitness centers to host on-site AEDs, while South Australia enforces similar requirements for public buildings by 2026. King County registers more than 5,000 devices linked to 911, bolstering survival odds when placement and dispatcher guidance converge. Yet bystander AED use appears in only 4% of out-of-hospital arrests, leaving a sizable adoption runway. Evidence from San Diego’s Project Heart Beat shows that municipal programs can raise access to levels mirroring the ubiquity of fire extinguishers, corroborating policy momentum.
Aging Workforce Training & Simulation Demand
As of 2024, the median age of American workers has risen to 44, a five-year increase since 2015.[3]U.S. FAA, “Draft Airworthiness Standards for Medical Payload Drones,” faa.gov In response, OSHA has begun recommending annual CPR refreshers. Moving away from traditional classroom drills, digital simulations are taking the lead. For instance, Laerdal’s QCPR Instructor app transmits compression metrics to cloud dashboards. This innovation has led to a 35% reduction in training costs per learner and an 18% increase in skill retention over six months, as highlighted in a 2025 peer-reviewed study. Furthermore, corporate buyers are increasingly opting for subscription-based e-learning packages. These packages not only align with workforce safety audits but also enhance institutional readiness and boost sales of external units.
Restraint Impact Analysis*
| RESTRAINT | (~) % IMPACT ON CAGR FORECAST | GEOGRAPHIC RELEVANCE | IMPACT TIMELINE |
|---|---|---|---|
| Stringent multi-region regulatory frameworks | −1.1% | Global, longest delays in EU & U.S. | Medium term (2-4 years) |
| High total cost of ICD implantation & follow-ups | −0.9% | APAC, MEA, Latin America | Long term (≥ 4 years) |
| Cyber-security risks for connected devices | −0.5% | North America & EU | Short term (≤ 2 years) |
| Lithium-supply pressure on batteries | −0.6% | Global, Asia-Pacific hubs most exposed | Medium term (2-4 years) |
| Source: ���ϲ����� | |||
Stringent Multi-Region Regulatory Frameworks
The EU Medical Device Regulation (MDR 2017/745) obliges recertification of legacy products, with half of manufacturers planning portfolio cuts and roughly one-third of devices slated for exit owing to cost and timeline burdens. ZOLL’s sequential MDR approvals for its AED line illustrate the added year or more now needed for market entry. Across the Atlantic the U.S. Food and Drug Administration requires software bills of materials and vulnerability reporting for “cyber devices,” adding documentation layers that elongate clearance cycles. Divergent rule sets compel dual certification efforts, straining budgets and slowing innovation throughput in the defibrillator market.
High Total Cost of ICD Implantation & Follow-Ups
Economic burden remains pronounced, especially in health systems lacking broad reimbursement. Italian registry data record mean EUR 5,662 hospitalization costs after generator replacement, with 9.6% of patients rehospitalized within a year. The EuroEco study found remote monitoring profitable for German and UK providers but loss-making in Belgium, Spain, and the Netherlands where payment codes lag. In Asia-Pacific, lower per-capita spend and insurance coverage suppress implant volumes despite rising clinical need, muting potential of the defibrillator market until affordability improves.
*Our updated forecasts treat driver/restraint impacts as directional, not additive. The revised impact forecasts reflect baseline growth, mix effects, and variable interactions.
Segment Analysis
By Product: Implantables Dominate, External Units Accelerate
In 2025, implantable systems dominated the defibrillator market, securing a commanding 71.02% share, particularly for patients with ejection fractions below 35%. While implantable defibrillators are set to grow at a steady 6.8% CAGR, the external segment is on a faster track, projected to expand at 7.64% through 2031. This surge is buoyed by regulatory support, notably the push for placing AEDs in every European workplace within three years. Transvenous ICDs are still divided between single- and dual-chamber models, but subcutaneous and extravascular designs are gaining traction by mitigating lead-related complications.
External defibrillators, while contributing a smaller base, are projected to compound at 7.64% through 2031, reflecting public-access expansion and AI-driven form factors. Wearable cardioverter units such as the FDA-cleared Jewel Patch enhance compliance by suppressing nuisance alarms and enabling barefoot therapy, factors that deepen user adoption. Drone-delivered automated external devices could raise survival by 34% in four-minute response scenarios, underscoring logistical innovation now shaping the defibrillator market. The segment also gains from subscription models that bundle connectivity, maintenance, and analytics, making budgeting predictable for municipalities and enterprises.
By End User: Home Care Gains as Remote Monitoring Matures
Hospitals and cardiac centers accounted for 77.60% of the defibrillator market in 2025, driven by procedural complexity and the concentration of electrophysiology expertise. In-facility use involves manual defibrillators and advanced resynchronization implants, which support intensive replacement cycles and capital budget allocation. Cloud platforms extend monitoring beyond discharge, enabling providers to bill for remote interrogation and trigger a services annuity that lifts spend per patient.
Home-care settings, though smaller, are scaling at a 7.98% CAGR as insurers back telemonitoring and patients seek autonomy. Studies show patients connected to St. Jude Medical’s Merlin network experienced 2.4 times higher survival, validating the value proposition for payers. South Korea’s first remote program reduced clinic visits by nearly 1 per patient annually while maintaining satisfaction above 90%, demonstrating operational feasibility. As AI-enabled wearables proliferate, daily monitoring becomes frictionless, expanding the defibrillator market in domiciliary environments.
By Patient Type: Pediatric Applications Expand with Miniaturized Leads
Adult patients accounted for 63.05% of the defibrillator market in 2025, reflecting the epidemiology of coronary artery disease and heart failure. Adoption patterns remain stable, but upgrades to newer extravascular systems should prompt a replacement wave over the forecast period. Leadless options also mitigate infection risks, adding incremental adult demand.
Pediatric deployments, though modest in number, are projected to grow 7.78% annually as miniaturization and battery advances align with unique anatomical needs. Successful extravascular ICD placement in patients as young as two years eliminates transvenous-lead challenges and positions the defibrillator market for durable pediatric growth. Research into left-bundle pacing and adaptive algorithms promises long-term cardiac support compatible with maturing physiology.
Geography Analysis
North America led the defibrillator market with 43.85% market share in 2025, supported by integrated emergency systems and reimbursement clarity. King County’s 5,000-plus registered AEDs linked to dispatch exemplify best-practice public-access integration. The FDA’s prompt clearance of innovations such as the Aurora EV-ICD and Jewel Patch cultivates early adoption and reinforces regional leadership. Drone pilots in North Carolina cut response times to 4 minutes, hinting at further survival gains once scaled.
Europe sustains moderate growth as MDR compliance stabilizes. Although roughly one-third of devices risk discontinuation, successful certifications, such as ZOLL’s AED line, demonstrate that committed manufacturers can navigate the process. Remote monitoring uptake remains uneven: Germany and the UK reimburse connectivity, whereas Belgium and Spain lag, tempering defibrillator market penetration. Netherlands-based drone-AED projects highlight technological enthusiasm, and mandated installations in South Australia mirror the regulatory push for accessibility.
Asia-Pacific exhibits the highest CAGR of 8.21%, driven by healthcare expenditure growth above OECD averages despite lower baseline spending. Training deficits are sizable only 17.5% of Chinese nurses feel AED-ready but national curricula and corporate upskilling initiatives aim to bridge gaps. ICD adoption still trails Western benchmarks due to cost constraints, yet expanding insurance coverage and local manufacturing e.g., MicroPort’s European catheter rollout should ease affordability and bolster the defibrillator market. Venture funding volatility poses a short-term challenge, but demographic shifts and policy support suggest durable demand through 2031.
Competitive Landscape
The defibrillator market is moderately consolidated, with incumbents leveraging R&D heft to maintain share. Medtronic commands leadership in extravascular systems; its Aurora EV-ICD hit 98.7% effectiveness, underscoring clinical edge.
Abbott capitalizes on dual-chamber leadless pacing, having secured the the CE mark for the AVEIR DR system, which achieves 97% AV synchrony, positioning the firm firmly in combined therapy domains. Boston Scientific’s modular mCRM platform validates wireless cardiac ecosystem strategy and opens cross-sell avenues between pacemakers and defibrillators.
Strategic transactions reshape portfolios: Johnson & Johnson integrated heart-pump maker Abiomed for USD 16.6 billion to broaden cardiovascular offerings, while Teleflex bought BIOTRONIK’s vascular intervention unit for USD 825 million, adding cath-lab synergies. New entrants exploit connectivity niches; ZOLL’s cloud analytics and Medtronic’s subscription services signal migration toward recurring revenue. Cybersecurity compliance now differentiates suppliers as FDA regulations require software bills of materials, pushing smaller players to partner or exit, and potentially raising barriers to entry.
Defibrillator Industry Leaders
Boston Scientific Corporation
Abbott Laboratories
Medtronic PLC
Koninklijke Philips NV
Nihon Kohden Corporation
- *Disclaimer: Major Players sorted in no particular order


Recent Industry Developments
- February 2026: ZOLL launched Zenix, a monitor-defibrillator approved under EU MDR.
- February 2026: BIOTRONIK released Acticor Sky and Rivacor Sky ICD/CRT-D family featuring the first CE-approved left bundle-branch pacing high-voltage device.
- February 2026: Edwin O’Sullivan Training Solutions began supplying AEDs to Irish schools, sports clubs, and community organizations.
- January 2026: Centre Hospitalier de l’Université de Montréal implanted BIOTRONIK’s Amvia Sky HF-T QP triple-chamber pacemaker following its full-market release.
- May 2025: Element Science gained FDA approval for the Jewel Patch wearable cardioverter defibrillator.
Global Defibrillator Market Report Scope
As per the scope of the report, a defibrillator is a life-saving device that helps deliver a therapeutic shock to a patient's heart in life-threatening conditions, such as ventricular fibrillation, cardiac arrhythmia, and pulseless ventricular tachycardia.
The defibrillator market is segmented by product, end user, and patient type. By product, the market is segmented into implantable cardioverter defibrillators (ICDs) and external defibrillators (EDs). The implantable cardioverter defibrillator (ICD) is segmented into transvenous implantable cardioverter defibrillator (T-ICD), subcutaneous implantable cardioverter defibrillator (S-ICD), and CRT with pacemaker and ICD function (CRT-D). The external defibrillator (ED) is segmented into automated external defibrillators (AEDs), manual external defibrillators, and wearable cardioverter defibrillators (WCDs). By end user, the market is segmented into hospitals, emergency care, home care, and other end users. By geography, the market is segmented into North America, Europe, Asia-Pacific, the Middle East and Africa, and South America. The market report also covers estimated market sizes and trends for 17 countries across major regions worldwide. The report offers market size and forecasts in value (USD) for the above segments.
| Implantable Cardioverter Defibrillator (ICD) | Transvenous ICDs (T-ICDs) | Single-Chamber |
| Dual Chamber | ||
| Subcutaneous ICDs (S-ICDs) | ||
| Cardiac Resynchronization Therapy-D (CRT-D) | ||
| External Defibrillator | Automated External Defibrillators (AEDs) | Semi Automated |
| Fully Automated | ||
| Manual External Defibrillators | ||
| Wearable Cardioverter Defibrillators (WCDs) | ||
| Hospitals & Cardiac Centers |
| Home Care Settings |
| Other End Users |
| Adult Patients |
| Pediatric Patients |
| North America | United States |
| Canada | |
| Mexico | |
| Europe | Germany |
| United Kingdom | |
| France | |
| Italy | |
| Spain | |
| Rest of Europe | |
| Asia-Pacific | China |
| India | |
| Japan | |
| Australia | |
| South Korea | |
| Rest of Asia-Pacific | |
| Middle East and Africa | GCC |
| South Africa | |
| Rest of Middle East and Africa | |
| South America | Brazil |
| Argentina | |
| Rest of South America |
| By Product | Implantable Cardioverter Defibrillator (ICD) | Transvenous ICDs (T-ICDs) | Single-Chamber |
| Dual Chamber | |||
| Subcutaneous ICDs (S-ICDs) | |||
| Cardiac Resynchronization Therapy-D (CRT-D) | |||
| External Defibrillator | Automated External Defibrillators (AEDs) | Semi Automated | |
| Fully Automated | |||
| Manual External Defibrillators | |||
| Wearable Cardioverter Defibrillators (WCDs) | |||
| By End User | Hospitals & Cardiac Centers | ||
| Home Care Settings | |||
| Other End Users | |||
| By Patient Type | Adult Patients | ||
| Pediatric Patients | |||
| By Geography | North America | United States | |
| Canada | |||
| Mexico | |||
| Europe | Germany | ||
| United Kingdom | |||
| France | |||
| Italy | |||
| Spain | |||
| Rest of Europe | |||
| Asia-Pacific | China | ||
| India | |||
| Japan | |||
| Australia | |||
| South Korea | |||
| Rest of Asia-Pacific | |||
| Middle East and Africa | GCC | ||
| South Africa | |||
| Rest of Middle East and Africa | |||
| South America | Brazil | ||
| Argentina | |||
| Rest of South America | |||


Key Questions Answered in the Report
How big is the Defibrillator Market?
The Defibrillator Market size is expected to reach USD 15.65 billion in 2026 and grow at a CAGR of 7.10% to reach USD 22.05 billion by 2031.
How fast will demand for external defibrillators grow?
External defibrillators are forecast to register a 7.64% CAGR between 2026 and 2031.
Who are the key players in Defibrillator Market?
Boston Scientific Corporation, Abbott Laboratories, Medtronic PLC, Koninklijke Philips NV and Nihon Kohden Corporation are the major companies operating in the Defibrillator Market.
Which is the fastest growing region in Defibrillator Market?
Asia Pacific is estimated to grow at the highest CAGR over the forecast period (2026-2031).
Which region has the biggest share in Defibrillator Market?
North America commanded 43.85% of global sales in 2025.
Page last updated on:









